Phase 1 - Expert consultation
- Definition of the project´s aim
- Thorough discussion of antigen characteristics and project requirements

Anti-drug antibodies (ADAs) may occur as an organism's response to a biotherapeutic (e.g. small oligonucleotides or antibodies) during treatment, a process also known as unwanted immunogenicity. ADAs harbor the risk of mediating unwanted biological or physiological effects, either by inhibiting the therapeutic effect of the treatment itself or by provoking adverse events.
The immunogenicity assessment of a therapeutic drug is based on a three-tiered approach and involves the use of anti-drug antibodies or anti-idiotypic antibodies as positive assay control. Since the thorough examination of the occurrence of ADAs in patient samples is particularly challenging, a multi-step approach is currently recommended by regulatory authorities. This multi-step approach is offered by BioGenes together with FyoniBio. Assay development and validation are conducted according to the FDA & EMA guidelines.
BioGenes offers the development of an immunogenicity ELISA for screening of anti-drug antibodies in serum samples by using a soluble bridging ELISA. In this set-up the ADA establishes a molecular bridge between the labeled drug antibodies. For assay development and as a comparative positive control, polyclonal anti-IDs specific for the drug antibody are used.
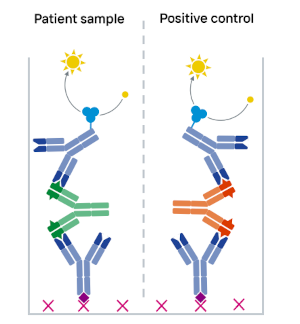
Screening-positive samples are subsequently analyzed in a competitive confirmation assay using the therapeutic drug antibody as competitor. Sufficient ADA signal inhibition by the drug verifies a sample as truly positive.
Finally, confirmed positive samples are analyzed in the semi-quantitative titration assay where the Log2 titer of a sample is determined from a 2-fold serial dilution. For positive samples, further characterization of the isotype and the neutralizing activity of the ADA might be performed.
Each project is conducted in close cooperation with the customer and consists of different work phases.
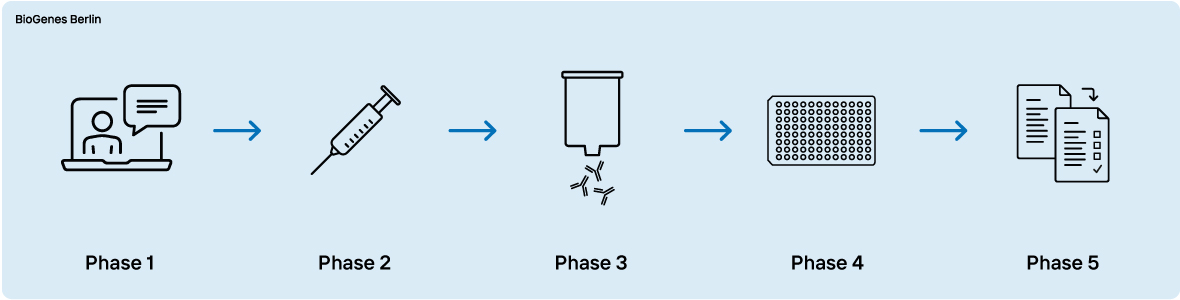
Phase 1 - Expert consultation
Phase 2 - Immunization and development of polyclonal antibodies
Phase 3 - Affinity purification
Phase 4 - Development and optimization of the bridging ELISA in solution
Phase 5 - Antibody & method transfer
Approximate project duration: 25-30 weeks
To provide you with an all-inclusive service, BioGenes works closely together with FyoniBio. By combining our scientific expertise, we can offer you an all-in-one package ranging from custom antibody and assay development to ELISA validation and the analysis of pre-clinical and clinical samples according to GCLP guidelines. However, customers can also choose to independently obtain the individual services from either company alone.
Contact
If you need more information, a specific offer
or want to talk to an expert
Newsletter
Keep me updated